Measurement of Matter
Based on Maharashtra Board Class 9-Science-Chapter-4
Solution
Question 1:
Give examples.
1. Positive ions
Positive ions : (i) Sodium Ions Na+ (ii) Silver ions Ag+ (iii) Aluminium ions Al3+ , (iv) Chromium ion Cr3+ (v) Calcium ion Ca2+
2. Basic radicals
Basic Radical : (i) Na+ (ii) Fe2+ (iii) Ag+ (iv) Al3+ (v) Cr3+ (vi) Sn2+.
3. Composite radicals
Composite Radical : (i) SO4 2- (ii) H3O+ (iii) NH4+ (iv) HCO3- (v) SO42- (vi) NO4-.
4. Metals with variable valency
Metal With Variable Valency : (i) Cu → Cu+ Cu2+, (ii) Hg → Hg+, Hg2+ (iii) Fe → Fe2+ , Fe3+.
5. Bivalent acidic radicals
Bivalent Acidic Radical : (i) S2- (ii) O2- (iii) Se2-
6. Trivalent basic radicals
Trivalent Basic Radical : (i) Al3+ (ii) Cr3+ (iii) Fe3+ (iv) Au3+.
Question 2:
Write symbols of the following elements and the radicals obtained from them and indicate the charge on the radicals.
Mercury, potassium, nitrogen, copper, sulphur, carbon, chlorine, oxygen
Element
Symbols
Charge
1. Mercury
Hg
Mercurous- Hg+
2. Potassium
K
Potassium- K+
3. Nitrogen
N
Azide- N3-
4. Copper
Cu
Cuprous- Cu+
5. Sulphur
S
Sulphide- S2-
Sulphate- SO42-
Sulphite- SO32-
6. Carbon
C
Carbide-C-
7. Chlorine
Cl
Chloride- Cl-
8. Oxygen
O
Oxide- O2-
Question 3:
Write the steps in deducing the chemical formulae of the following compounds.
(i) Sodium sulphate,
Sodium sulphate : Step 1 : Write the symbols and valencies of the radicals. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula of Sodium sulphate is Na2SO4
Symbols
Na
SO4
Valency
1
2

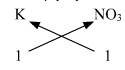
(ii) Potassium nitrate,
Potassium nitrate : Step 1 : Write the symbols and valencies of the radicals. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula of Potassium nitrate is KNO3
Symbols
K
NO3
Valency
1
1
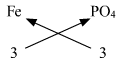
(iii) Ferric phosphate
Ferric phosphate : Step 1 : Write the symbols and valencies of the radicals. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula of Ferric phosphate is FePO4
Symbols
Fe
PO4
Valency
3
3
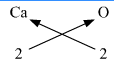
(iv) Calcium oxide
Calcium oxide : Step 1 : Write the symbols and valencies of the radicals. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula of Calcium oxide CaO
Symbols
Ca
O
Valency
2
2
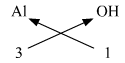
(v) Aluminium hydroxide
Aluminium hydroxide : Step 1 : Write the symbols and valencies of the radicals. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula of Aluminium hydroxide Al(OH)3
Symbols
Al
OH
Valency
3
1
Question 4:
Write answers to the following questions and explain your answers.
1. Explain how the element sodium is monovalent.
Sodium is a metal which has atomic number 11. It has 1 electron in its last shell which means it has 1 valence electron. In order to get stability by acquiring nearest noble gas configuration. Sodium prefers to loose 1 electron. Hence, we say that sodium has a valency of one and is thus monovalent.
2. M is a bivalent metal. Write down the steps to find the chemical formulae of its compounds formed with the radicals, sulphate and phosphate.
M is a bivalent metal that is : M2+ Step 1 : Write the symbols and valencies of of M and sulphate. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula is MSO4 M is a bivalent metal that is : M2+ Step 1 : Write the symbols and valencies of of M and Phosphate. Step 2 : Cross-multiply symbols of radicals with their respective valency. Step 3 : Write down the chemical formula of the compound. The chemical formula is M3(PO4)2
Symbols
M
SO4
Valency
2
2

Symbols
M
PO4
Valency
2
3

3. Explain the need for a reference atom for atomic mass. Give some information about two reference atoms.
4. What is meant by Unified Atomic Mass.'
Instead of relative atomic mass, it is now possible to determine atomic masses in kg. The Unified Atomic Mass Unit or Dalton is a standard unit of mass that quantifies mass on an atomic or molecular scale.
5. Explain with examples what is meant by a 'mole' of a substance.
A mole is that quantity of a substance whose mass in grams is equal in magnitude to the molecular mass of that substance in daltons. The SI unit is mole. Examples The molecular mass of oxygen is 32 u; 32 g oxygen is 1 mole of oxygen. The atomic mass of carbon is 12 u; 12 g carbon is 1 mole of carbon. The molecular mass of H2O is 18 u; 18 g H2O is 1 mole of water. Number of moles of Mass of substance (n) = \(\frac{\text{Mass of substance in grams}}{Molecular mass of substance}\)
Question 5:
Write the names of the following compounds and deduce their molecular masses.
(i) Na2 SO4 ,(ii) K2 CO3 ,(iii) CO2 ,(iv) MgCl2 ,(v) NaOH, (vi) AlPO4 ,(vii) NaHCO3
(i) Na2SO4 - Sodium sulphate. Molecular mass= sum of masses of individual components Atomic mass of, Na = 23u, S=32u, O=16u ∴ Molecular mass of Na2SO4 = 2x(23) + 32 + 4x(16)= 142u (ii) K2CO3 - Potassium carbonate. Atomic mass of, K=39u, C=12u, O=16u ∴ Molecular mass of K2CO3 = 2x(39) + 12 + 3x(16) = 138u (iii) CO2 - Carbon dioxide. Atomic mass of, C=12u, O=16u ∴ Molecular mass of CO2 = 12 + 2x(16) = 44u (iv) MgCl2 - Magnesium chloride. Atomic mass of, Mg=24u, Cl=35.5u ∴ Molecular mass of MgCl2 = 24 + 2x(35.5) = 95u (v) NaOH - Sodium hydroxide. Atomic mass of, Na=23u, O=16u, H=1u ∴ Molecular mass of NaOH = 23 + 16 + 1 = 40u (vi) AlPO4 - Aluminium phosphate Atomic mass of, Al= 27u, P=31u, O=16u ∴ Molecular mass of AlPO4 = 27 + 31 + 4x(16) = 122u (vii) NaHCO3 - Sodium bicarbonate Atomic mass of, Na=23u, H= 1u, C=12u, O=16u ∴ Molecular mass of NaHCO3 = 23 + 1 + 12 + 3x(16) = 84u
Question 6:
Two samples ‘m’ and ‘n’ of slaked lime were obtained from two different reactions. The details about their composition are as follows:
‘sample m’ mass : 7g
Mass of constituent oxygen : 2g
Mass of constituent calcium : 5g
‘sample n’ mass : 1.4g
Mass of constituent oxygen : 0.4g
Mass of constituent calcium : 1.0g
Which law of chemical combination does this prove? Explain.
Slaked lime have two samples ‘m’ and ‘n’ Sample m Mass :7g In sample m, the ratio of proportion of elements (calcium:oxygen) is 5:2 Sample n Mass :1.4g In sample n, the ratio of proportion of elements (calcium:oxygen) is 1.0:0.4 Out of all the laws of chemical combination, this is proved by "Law Of Constant Proportion". Law Of Constant Proportion states that "The proportion by weight of the constituent elements in various samples of compound is fixed in ratio". On simplifying the ratio proportion by mass, we get the same values which verifies "The Law Of Constant Proportion".
Mass of constituent oxygen :2g
Mass of constituent calcium :5g
Ca:O =5:2 by wight
Mass of constituent oxygen :0.4g
Mass of constituent calcium:1.0g
Ca:O =1.0:0.4 by weight i.e.
=10:4
=5:2
Question 7:
Deduce the number of molecules of the following compounds in the given quantities.
(i) 32g oxygen, (ii) 90g water, (iii) 8.8g carbon dioxide, (iv) 7.1g chlorine.
(i) 32 g of oxygen : Number of moles of O2 = Mass of O2 in grams/Molecular mass of O2 = 32/32 = 1mol (ii) 90 g of water : Number of moles in water = Mass of water in grams/Molecular mass of water = 90/18 = 5mole ∴ 5 moles of H2O contains-----5 x 6.022 x 1023 molecules = 30.11 x 1023 molecules ∴ 90g of water contains 30.11 x 1023 molecules (iii) 8.8 g of CO2 : Molecular mass of Carbon dioxide CO2 =12+2x(16) =44 ∴0.2 moles of CO2 contains-----0.2 x 6.022 x 1023 molecules = 1.2044 x 1023 molecules ∴8.8 g of CO2 contains 1.2044 x 1023 molecules (iv) 7.1 g of chlorine ∴0.1 moles of Cl2 contains-----0.1 x 6.022 x 1023 molecules = 0.6022 x 1023 molecules ∴ 7.1 g of chlorine contains 0.6022 x 1023 molecules
Molecular mass of O2 = 2x16=32
∴ 1 mole of O2 contains-----6.022 x 1023 molecules
∴ 32g of oxygen contains 6.022 x 1023 molecules
Molecular mass of water H2O= 2x(1)+16 =18
Number of moles in CO2 = Mass of CO2 in grams / Molecular mass of CO2 = 8.8/44 = 0.2mol
Molecular mass of Chlorine Cl2 = 2x(35.5)=71
No. of moles in chlorine=Mass of chlorine in grams/Molecular mass of chlorine =7.1/71 = 0.1mol
Question 8:
If 0.2 mol of the following substances are required how many grams of those substances should be taken?
(i) Sodium chloride, (ii) magnesium oxide, (iii) calcium carbonate
(i) We know that, Molar mass= sum of constituent atomic masses Molar mass of NaCl= 23 + 35.5 = 58.5 g/mol Number of moles of a substance= \(\frac{\text{Molar mass of substance in grams}}{\text{Molecular mass of the substance}}\) 0.2=\(\frac{x}{58.5}\) x = 0.2×58.5 = 11.7g We need, 11.7 g of NaCl for obtaining 0.2 moles of NaCl. (ii) Molar mass of MgO= 24 + 16 = 40 g/mol Number of moles of a substance= \(\frac{\text{Molar mass of substance in grams}}{\text{Molecular mass of the substance}}\) 0.2=\(\frac{x}{40}\) x = 0.2×40 = 8g We need, 8 g of MgO for obtaining 0.2 moles of MgO. (iii) Molar mass of CaCO3= 40 + 12 + 3 (16) = 100 g/mol Number of moles of a substance= \(\frac{\text{Molar mass of substance in grams}}{\text{Molecular mass of the substance}}\) 0.2=\(\frac{x}{100}\) x =0.2×100= 20 g We need, 20 g of CaCO3 for obtaining 0.2 moles of CaCO3.
Useful links :
| Main Page : - Maharashtra Board Class 9th Science & Technology - All chapters notes, solutions, videos, test, pdf.
Books : MSBSHSE -Class 9th Science Text Books – Chapter wise PDF for download Previous Chapter : Chapter 3: Current Electricity - view online Solution Next Chapter : Chapter 5- Acids, Bases and Salts -view online Solution |
